
Diagnostics with traceable QC.
Clinical chemistry, immunoassay, and point-of-care testing programs can be documented with lot traceability, Levey-Jennings QC, Westgard rule review, and LIS bidirectional interfaces.

Clinical chemistry, immunoassay, and point-of-care testing programs can be documented with lot traceability, Levey-Jennings QC, Westgard rule review, and LIS bidirectional interfaces.

Connected observations can be routed into clinical workflows with HL7 v2, FHIR observation resources, escalation thresholds, and audit-ready cybersecurity controls.
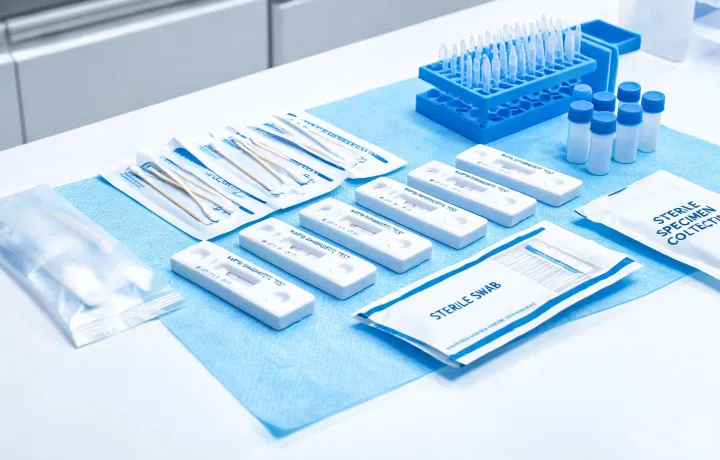
Reagent, cartridge, and infection-control supplies are mapped to par levels, cold-chain events, UDI traceability, and standard operating procedures for multi-site procurement teams.
| Requirement | Abbott planning answer |
|---|---|
| Throughput | Batch, random-access, and POCT profiles mapped to tests per hour and TAT percentile. |
| LIS | HL7 v2.5.1 ORM/ORU, ASTM E1394, and FHIR Lab IG interface planning. |
| Validation | Precision, linearity, reportable range, and method comparison packages. |
| Requirement | Abbott planning answer |
|---|---|
| Data model | FHIR Observation resources with role-based access and audit logging. |
| Cybersecurity | MDS2, SBOM, coordinated disclosure, and CVE response SLA templates. |
| Escalation | Smart threshold routing for care coordinators and biomedical engineering. |
| Requirement | Abbott planning answer |
|---|---|
| UDI | GS1 UDI-DI traceability for stocked reagents, kits, and consumables. |
| Cold chain | Temperature excursion logs and lot-release escalation paths. |
| Contracts | GPO, multi-site replenishment, and utilization-based ordering models. |
Central chemistry, immunoassay, and rapid-response testing with CAP-ready QC.
High-volume sample handling, middleware routing, and reagent economics.
Monitoring workflows that blend wearables, escalation rules, and EHR documentation.
Remote observation capture with HIPAA-safe patient engagement pathways.
Rapid diagnostics and consumables tied to lot traceability and outbreak response.

Ask for a demo packet with service tiers, interface assumptions, validation documentation, and consumables planning inputs.
Request the Packet